My question is: how do you calculate the amount of energy required to maintain a water temperature of 100 degrees C over a period of 5 mins. I have looked into .... Mar 16, 2020 — how do you calculate the energy needed to heat water? Calculate the kilowatt-hours (kWh) required to heat the water using the following .... Dec 27, 2015 — One liter of water is about 1 Kg or 1000g, giving Q = m x (delta T) x Specific heat of water = 1000g x 80C x 1 cal/g C= 80,000 cal or 80 Kcals. Technically, 80 kcals ...
For instance, you can check how much heat you need to bring a pot of water to the ... The specific heat capacity is the heat or energy required to change one unit .... Since Cp is measured in pounds, your mass of water heated should be measured in pounds as well. Thus, if you only know the number of gallons, you must .... May 16, 2018 — Calculate Kilowatt-Hours. Calculate the kilowatt-hours (kWh) required to heat the water using the following formula: Pt = (4.2 × L × T ) ÷ 3600.. The specific heat capacity refers to the amount of heat required to cause a unit of ... But how much energy would be required to cause such a change of state? ... effort to calculate the quantity of heat required to change 50.0 grams of water from ...
energy required to boil water calculator
energy required to boil water calculator, how much energy to boil water calculator, how much energy is required to boil water, how much energy is needed to boil water Little girls 11, 020 @iMGSRC.RU
Calculator for the energy required to heat a water flow from a given temperature to a desired temperature, including the costs with gas and electrical heating.. Since the specific heat of water is 4.18J/g/°C we can calculate the amount of energy needed by the expression below. Energy required = 4.18 J/g/°C X 100g X 73° .... May 2, 2021 — What is the heat in Joules required to convert 25 grams of -10 °C ice into 150 °C steam? Useful information: heat of fusion of water = 334 J/g heat .... c = specific heat capacity of water, which is 1 cal/(g °C) · m = mass of water; i.e. 1 kg · T = the room temperature in °C · Δhvap = specific enthalpy of vaporization of .... It depends on how much water you have, and to what temperature you want to raise it to. Explanation: The equation for the amount of thermal ... Sexys, 934010_1419047644974264_12454269 @iMGSRC.RU
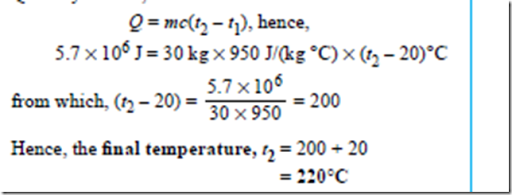
how much energy is required to boil water
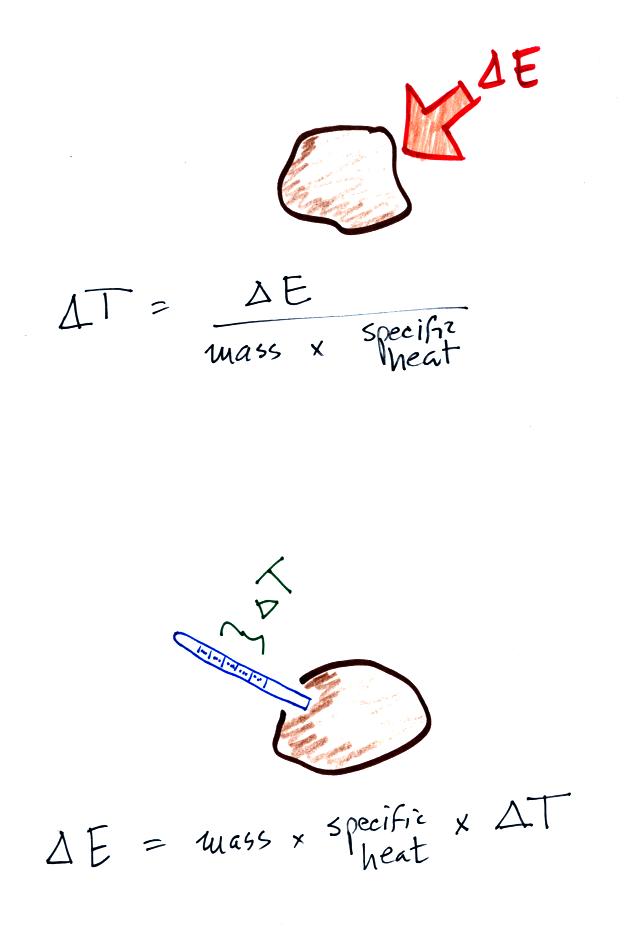
Water heater power P(kW) in kW is equal to 4.2 times of water qty L in Liters and the temperature difference divided by 3600. P(kW) = 4.2 x L x (T2-T1) / 3600.. A calorie is defined as the amount of heat required to change the temperature of one gram of liquid water by one degree Celsius (or one degree Kelvin).. Apr 29, 2020 — The mass of the water is 54.1 g and it has a specific heat of 4.18 J/g*°C. Calculate the energy required to heat a beaker of water at 33 C to boiling.. Calculating the wattage requirements to heat a system is a straightforward process as ... A BTU is the amount of energy required to heat 1 pound of water by 1°F ... TopSounds-Dark-Nights-Omnisphere-Bank-

Boiling time of water can be calculated with the volume, energy, efficiency, start and end temperature. Use our online calculator to find how long time needed to .... How do you calculate the energy needed to boil water? — How do you calculate the energy needed to boil water? 1 Answer. c = .... Mar 13, 2016 — The calculators on this page compute how long it takes to heat water, how much energy is consumed, and how much heating power is required .... Calculator that tells you how long it takes to heat water from start to end temperature with a given ... Other water heating calculators are: power and energy. 3e88dbd8be RUS Crystar
